Company tel:+86-379-63184520
Contact number:+86-15937921751
Postcode:471000
Email:info@yuxindiamond.com
Address:3-1-508 Luoyang National University Science Park, No. 2 Penglai Road, Jianxi District, Luoyang, China 471000
Nanodiamond for High Performance Phenolics Sensing
Phenolic compounds, a class of chemicals containing one or more hydroxyl groups attached to an aromatic ring, are ubiquitous in the environment. They occur naturally in plants and form an essential part of the human diet including wine and fruits. Also of artificial origin, these compounds are used in the manufacturing industry and are found in wastewaters, exhaust gases and as by-products of chemical engineering processes. Their occurrence in waterbodies is undesirable, as they can cause severe adverse effects on animals and humans above a specific dose, resulting in severe kidney, liver and cardiovascular diseases. At least phenolic compounds are listed by the US Environmental Protection Agency and the European Union as priority pollutants. Rapid and accurate monitoring of phenolic compounds in solution would be beneficial to quantifying and reducing the exposure to these contaminants.
In this work, we demonstrate that both nanodiamond and amorphous nanocarbon offer a facile solution to the aforementioned challenges due to their consistently high sensitivities, prevention from passivation and fouling and superior biocompatibility to enzyme modification. We provide a comprehensive analysis of their electrochemical performance and compare them to graphene-modified electrodes for detection of mono and biphenols. The wide range of electrochemical methods, including voltammetry and amperometry allow the comparison of three distinct carbon materials systematically and provide sensing performance benchmarks. Further, we demonstrate the simultaneous detection of co-existing phenolic isomers using nanocarbon electrodes, and apply these methods to detect phenolics in real Thames river water samples and green tea.
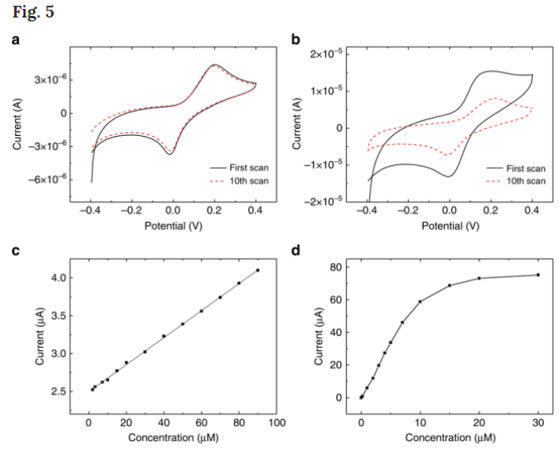
Fouling of electrodes is a problem for phenolic sensors35. The by-products from phenolic compound oxidation react with the electrode, thereby inactivating it. It is well-known that boron-doped diamond benefits from high chemical resistance22. To test whether nanodiamond-GCE resists fouling when used to detect phenolics, we performed cyclic voltammetry in HQ for consecutive cycles. After ten scan cycles (Fig. 5a in red) the voltammogram did not change significantly. In contrast, a dramatic drop of redox peak current was observed at a graphene electrode (Fig. 5b) indicating surface fouling. This comparison shows the excellent inertness of nanodiamond against phenolic compounds. We observed that peak currents at nanodiamond-GCE scale linearly with HQ concentration up to 100 μM (Fig. 5c). This shows that nanodiamond-GCE have a wider detection range. In comparison, graphene-GCE depart from linearity at concentrations above 10 μM (Fig. 5d), as well as nanocarbon-GCE (Supplementary Figure 4). We attribute fouling of graphene and nanocarbon electrodes to their sp2 carbon–carbon hybridisation, which facilitates the adsorption and reaction of phenolics oxidation by-products.
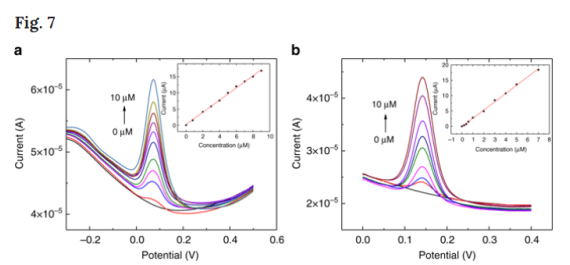
We used single-use nanocarbon-GCE to detect HQ and catechol in Thames river water samples. Known concentrations of HQ and catechol were added to river water samples (Fig. 8a in red) and SWV was used at nanocarbon-GCE to measure their concentration (Fig. 8a in blue). Found concentrations were extrapolated using the linear relationship shown in Fig. 7. Each measurement was repeated ten times. The average recovery rate was 102% for HQ and 101% for catechol. This experiment demonstrates that nanocarbon-GCE can be successfully applied in river water samples, without significant interference from additional co-existing compounds.
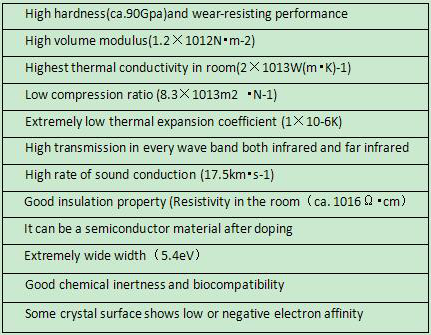
Diamond, as one of the most special materials in natural world, is featured with the highest hardness, low friction coefficient, high elasticity modulus, high thermal conductivity, high insulation class, wide energy gap, great sound propagation rate and favorable chemical stability, which are presented in below Table. In spite of such unique features, the natural diamond has always been existed in the form of gem, with its variability and rareness sharply limiting its application. Luoyang Yuxin Diamond Co., Ltd‘s CVD Diamond film, on the other hand, integrates such physical and chemical properties, with lower cost than natural diamond and applicable to be made into various shapes, thus enjoying extensive application prospect in electronic industry, optical field and mechanical industry.